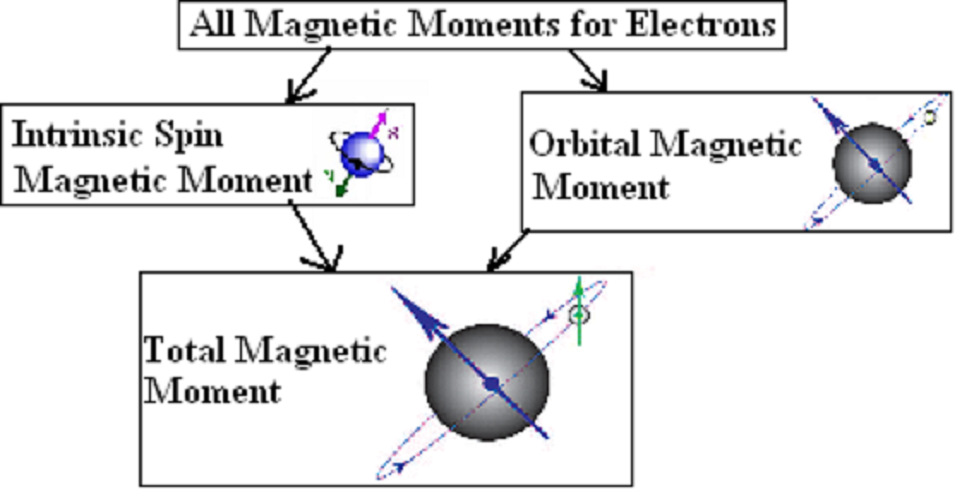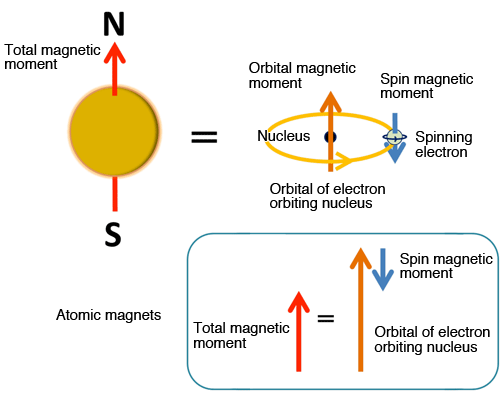
High-Accuracy Magnetic Property Measurement Method by Separating Spin and Orbital Magnetic Moments (Press Release) — SPring-8 Web Site

Physics - Ch 66.5 Quantum Mechanics: The Hydrogen Atom (34 of 78) Magnetic Moment in Hydrogen - YouTube

The magnetic moment of `Mg-\"atom\"` is more than `K-\"atom\"` as the former has two electrons in - YouTube

quantum mechanics - Ignoring spin, what is its orbital magnetic moment of an electron in a hydrogen atom in the 2p orbital? - Physics Stack Exchange

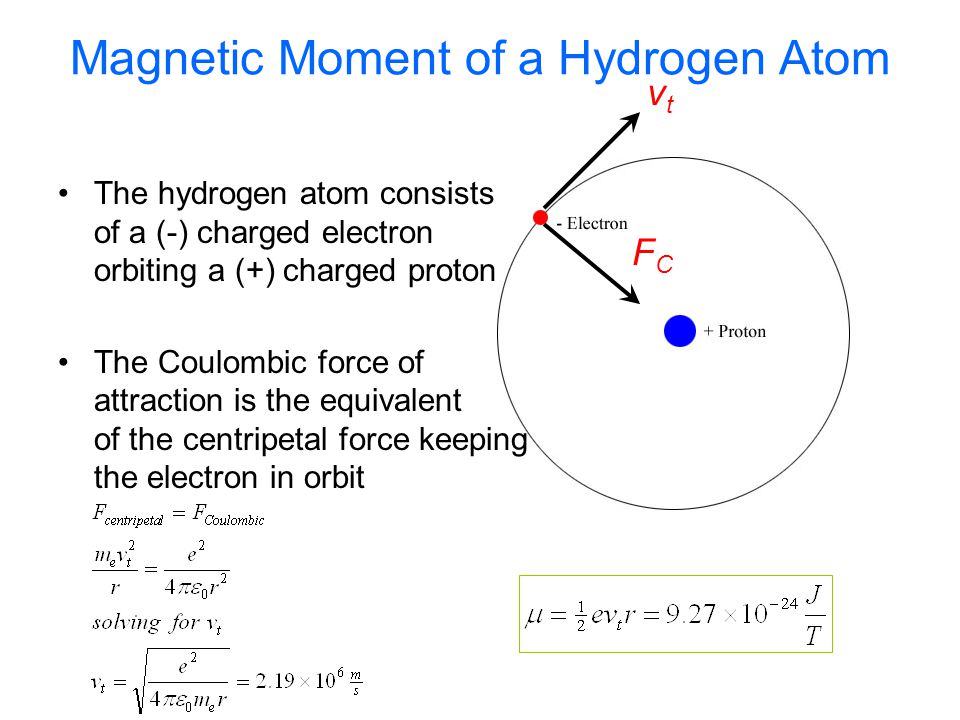
The electron in the hydrogen atom is moving with a speed of 2.3 x 10^6 m/s in an orbit of radius 0.53 A. Calculate the magnetic moment of the revolving electron. - Sahay Sir

SOLVED:The magnetic dipole moment of the iron atom is about 2.1 \times 10^{-23} \mathrm{A} \cdot \mathrm{m}^{2} . (a) Calculate the maximum magnetic dipole moment of a domain consisting of 10^{19} iron atoms. (


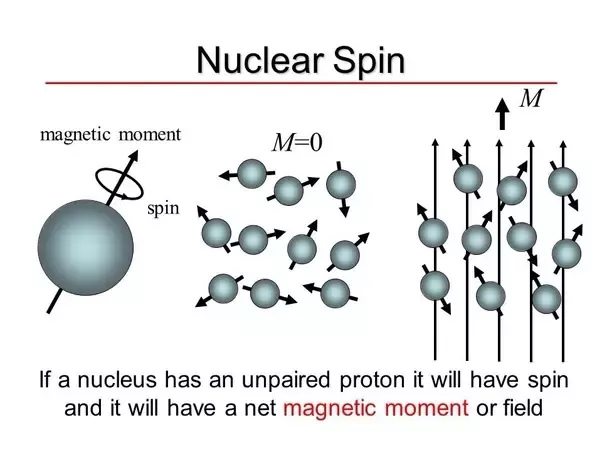


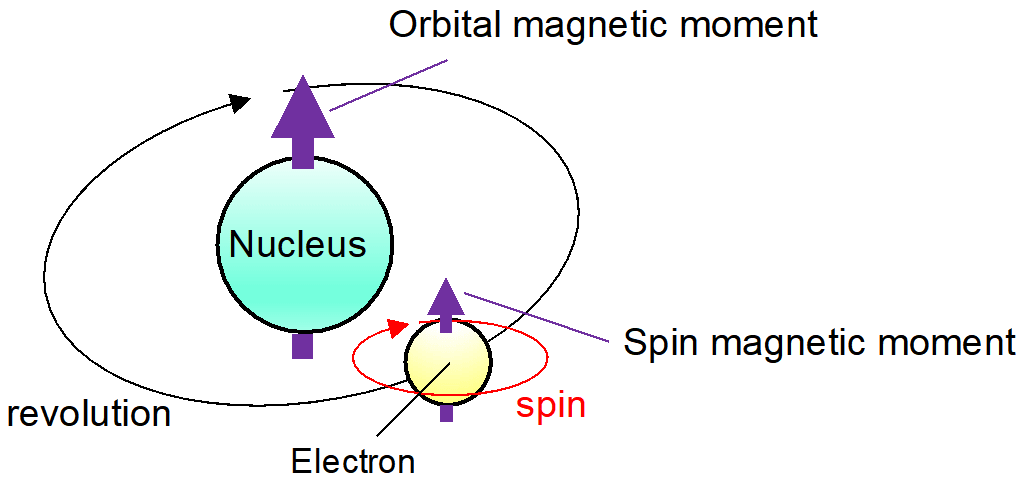
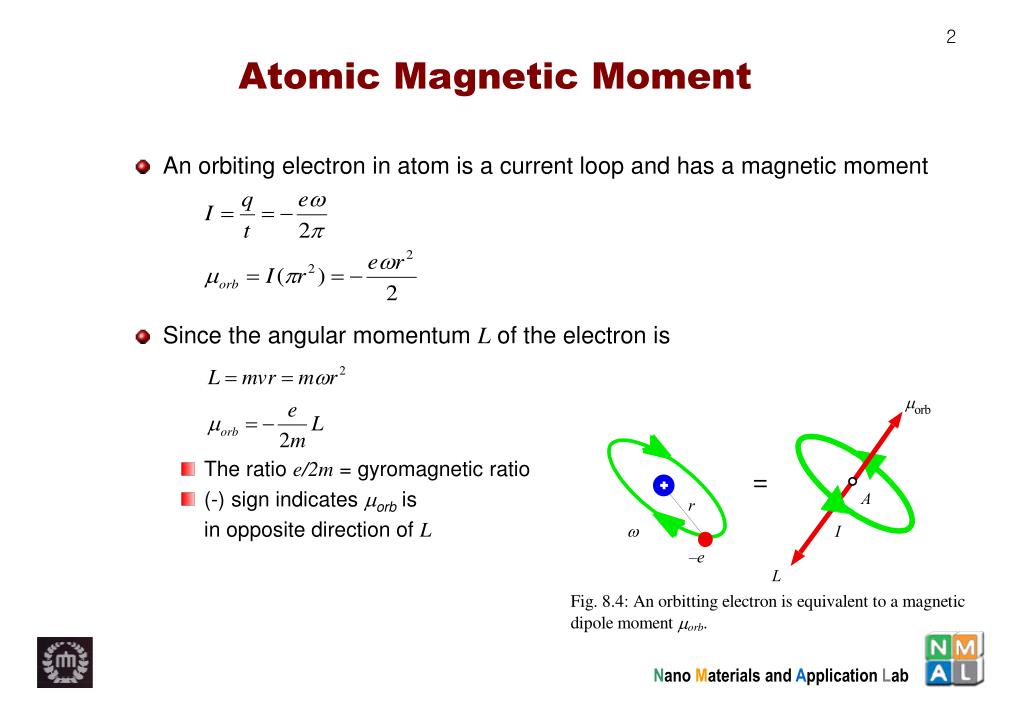





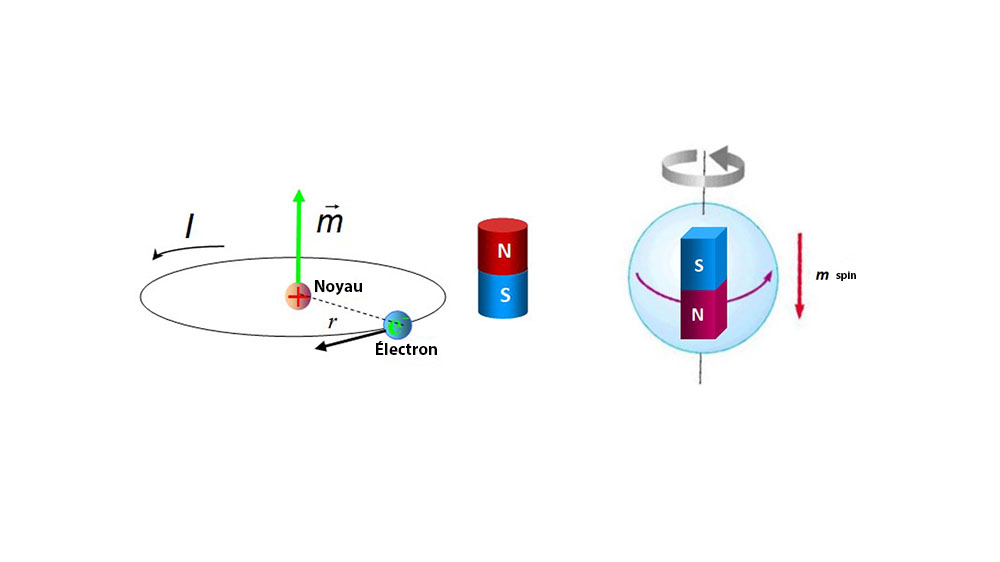


.PNG)