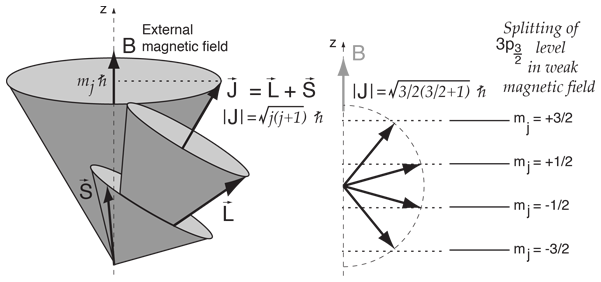
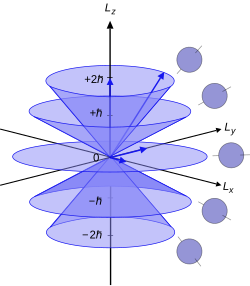
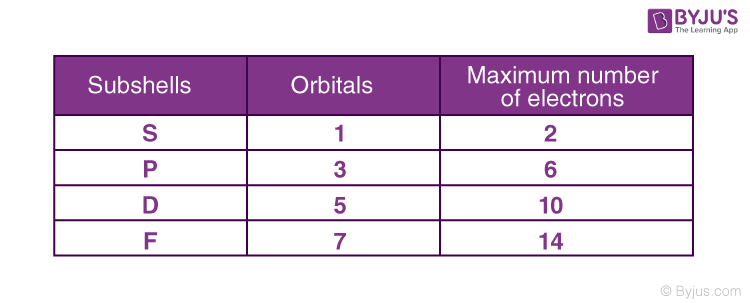
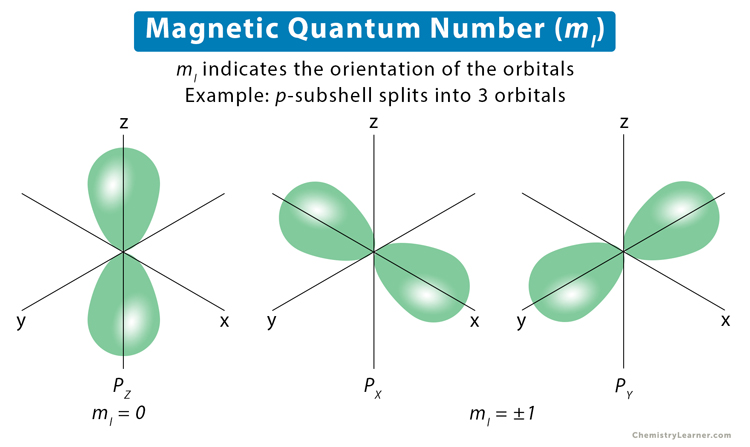
If the value of principal quantum number is 3, the total possible values for magnetic quantum number will be?

Total Angular Momentum L, L z, S, S z J and J z are quantized Orbital angular momentumSpin angular momentum Total angular momentum. - ppt download

The total number of possible values of magnetic quantum number for the value of l = 3 is | 11 | ... - YouTube

If value of azimuthal quantum number `l` is `2`, then total possible values of magnetic quantum - YouTube